
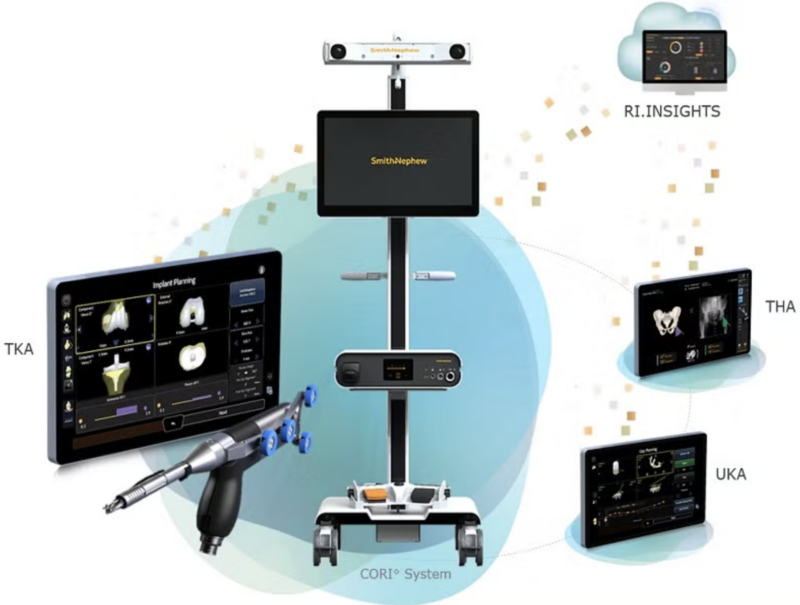
CORI™ Surgical System: Robotic-Assisted Knee Replacement Technology
The CORI™ Surgical System, developed by Smith+Nephew, is an advanced handheld robotic-assisted platform used in total, partial and revision knee replacement surgery. This technology is designed to help surgeons perform knee replacements with greater precision and personalization, supporting improved comfort, alignment and long-term outcomes for patients.
Discover the benefits of a Personalized Knee Replacement Surgery with the CORI™ Surgical System.
Introducing game-changing technologies that can help you start living your full life again—a more normal feeling knee,1 a smoother recovery2,3 and implants built to last. Call to request an appointment with Dr. Shannon at Orthopedics and Surgical Specialists
How the CORI™ System Works
Unlike some robotic systems that require a pre-operative CT scan or MRI, the CORI™ system is CT-free. Instead, it creates a detailed 3D digital model of your knee during surgery, based on your unique anatomy.
Handheld Robotic Precision
The surgeon uses a handheld robotic device that provides tactile guidance and extremely accurate bone shaping. This helps ensure bone removal is performed with sub-millimeter precision according to the surgical plan.
Personalized Surgical Planning
Before any permanent bone changes are made, the CORI™ system allows the surgeon to:
- “Test fit” the implant
- Balance ligaments
- Adjust alignment for optimal stability
This individualized approach helps tailor the procedure to each patient’s specific knee structure.
Versatile and Advanced Applications
CORI™ is one of the first robotic systems in the U.S. approved for revision knee replacement surgery, and it can also assist with hip navigation in certain cases.
Potential Benefits for Patients
Robotic-assisted surgery with the CORI™ system allows surgeons to combine their expertise with advanced technology to create a highly customized surgical plan — helping patients achieve a more comfortable, stable and confident recovery. Patients treated with CORI™-assisted knee replacement may experience:
Faster Recovery
Some patients return to daily activities up to six months sooner compared with traditional approaches.
Less Tissue Trauma
More accurate bone removal may help preserve natural structures, including important ligaments.
More Natural Knee Feel
Many patients report improved comfort, less pain and a knee that feels more normal during movement.
No Added Radiation
Because CORI™ does not require a pre-surgical CT scan, patients avoid unnecessary radiation exposure.
Long-Lasting Implant Support
The system is often paired with durable implant materials such as OXINIUM™, Oxidized Zirconium. This unique metal alloy is designed to reduce the effects of implant wear and tear. In fact, OXINIUM knee implants have been lab-tested for durability 9-times longer than the international standard for joint replacements.
What Makes CORI™ Different?
The CORI™ Surgical System uses advanced image-free technology, eliminating the need for a pre-surgical CT scan or MRI by mapping your knee in real time during the procedure to create a detailed 3D model for precise implant placement. During surgery, your surgeon remains fully in control while using a handheld robotic tool that provides tactile feedback and sub-millimeter accuracy for highly controlled bone preparation. The system also allows implant positioning and ligament balance to be assessed before any permanent bone changes are made, supporting a more personalized approach that may improve alignment, stability and overall comfort after surgery.
Questions to Ask Before Choosing the CORI™ Surgical System
If you’re considering knee replacement with the CORI™ Surgical System, these key questions can help guide your discussion with your surgeon:
- Am I a good candidate for robotic-assisted knee replacement?
- How does CORI™ improve precision compared to traditional surgery?
- What benefits can I realistically expect with this technology?
- What are the risks or potential complications?
- What will my recovery timeline look like?
At Orthopedics and Surgical Specialists, we believe informed patients make confident decisions. Our experienced team is committed to providing expert guidance, personalized recommendations and the highest standard of orthopedic care to help you choose the treatment that best supports your long-term mobility and success.
Important Safety Information
As with any medical treatment, individual results may vary. The performance of knee replacements depends on age, weight, activity level and other factors. There are potential risks and recovery takes time. People with conditions limiting rehabilitation should not undergo this surgery. Only an orthopaedic surgeon can determine if knee replacement surgery is an option for you.
The information listed on this site is for informational and educational purposes and is not meant as medical advice. Every patient’s case is unique and each patient should follow his or her doctor’s specific instructions. Please discuss nutrition, medication and treatment options with your doctor to make sure you are getting the proper care for your particular situation.
◊ Trademark of Smith+Nephew. The information on this site is intended for US residents only © 2022 Smith+Nephew
Smith+Nephew Facebook Page | Follow Smith+Nephew on Twitter | Privacy & Cookies | Terms of Use
References
- Hall, et al. Unicompartmental knee arthroplasty (alias uni-knee): an overview with nursing implications. Orthopaedic Nursing. 2004;23(3):163-171. Accessed April 25, 2019.
- Based on pre-surgical pain levels in UKA patients.
- Mayman DJ, Patel AR, Carroll KM. Hospital related clinical and economic outcomes of a bicruciate knee system in total knee arthroplasty patients. Poster presented at: ISPOR Symposium; May 19-23, 2018; Baltimore, Maryland, USA.
- Nodzo SR, Carroll KM, Mayman DJ. The Bicruciate Substituting Knee Design and Initial Experience. Techniques in Orthopaedics. 2018;33(1):37-41
- Compared to non-JOURNEY II knees; Based on BCS evidence
- 1Short-term Range of Motion is Increased after TKA with an asymmetric bicruciatestabilized implant.AcceptedPoster Presentation, AAOS 2018 New Orleans. Kaitlin M. Carroll, Peter K. Sculco, Brian CMichaels,RichardL. Murphy, Seth A, Jerabek, David J. Mayman
- 2J Orthop. 2017 Jan 7;14(1):201- 206. doi: 10.1016/j.jor.2016.12.005. eCollection 2017. Bi-cruciate substituting total knee arthroplasty improved medio-lateral instability in mid-flexion range
- In Vivo Kinematic Comparison of a Bicruciate Stabilized Total Knee Arthroplasty and the Normal Knee Using Fluoroscopy Trevor F. Grieco, MS a, *, Adrija Sharma, PhD a, Garett M. Dessinger, BS a, Harold E. Cates, MD b, Richard D. Komistek, PhD. The Journal of Arthroplasty, September 2017
- Testing concluded at 45 million cycles, ISO 14242-1 and 14243-3 define test completion at 5 million cycles. The results of laboratory wear simulation testing have not been proven to predict actual joint durability and performance in people. A reduction in wear alone may not result in improved joint durability and performance because other factors, such as bone structure, can affect joint durability and performance and cause medical conditions that may result in the need for additional surgery. These other factors were not studied as part of the testing.
- Iriuchishima T, Ryu K. Bicruciate substituting total knee arthroplasty improves stair climbing ability when compared with cruciate-retain or posterior stabilizing total knee arthroplasty. Indian J Orthop. 2019. doi:10.4103/ortho.IJOrtho_392_18.
- Smith JR, Picard F, Lonner J, et al. The accuracy of a robotically-controlled freehand sculpting tool for unicondylar knee arthroplasty. Congress of the International Society of Biomechanics. August 4-9, 2013. Natal, Brazil.
- Zardiackas, Lyle D., Kraay, Matthew J., Freese, Howard L, editors. Titanium, Niobium, Zirconium, and Tantalum for Medical and Surgical Applications ASTM special technical publication; 1471. Ann Arbor, MI: ASTM, Dec. 2005
